 | Chemical Periodicity
Examples of
Multiple Choice Questions
|
1.- The atom having the valence-shell configuration 4s2 4p5 would be in:
- (a) Group VIA and Period 5
- (b) Group IVB and Period 4
- (c) Group VIB and Period 7
- (d) Group VIIA and Period 4
- (e) Group VIIB and Period 4
- 2.
- Select the term best describing the series of elements: Mn, Fe, Co, Ni, Cu.
- (a) d-transition metals
- (b) representative elements
- (c) metalloids
- (d) alkaline earth metals
- (e) halogens
- 3.
- Which element has the largest atomic radius?
- (a) Li
- (b) Na
- (c) Rb
- (d) F
- (e) I
- 4.
- Which of the following terms accurately describes the energy associated with the process:
Li(g)
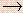 Li+(g) + e-
Li+(g) + e-
- (a) electron affinity
- (b) binding energy
- (c) ionization energy
- (d) electronegativity
- (e) none of these
- 5.
- The species that contains 24 protons, 26 neutrons and 22 electrons would be represented by the symbol:
- (a) 50V3+
- (b) 26Cr2+
- (c) 50Cr2+
- (d) 50Mn2+
- (e) none of these
- 6.
- Which element has the lowest first ionization energy?
- (a) He
- (b) Ne
- (c) Ar
- (d) Kr
- (e) Xe
- 7.
- Which element has the highest first ionization energy? (Note: this is an exception to the general trend - see notes- you may be responsible for this.)
- (a) Be
- (b) B
- (c) C
- (d) N
- (e) O
- 8.
- Which of these isoelectronic species has the smallest radius?
- (a) Br-
- (b) Sr2+
- (c) Rb+
- (d) Se2-
- (e) They are all the same size because they have the same number of electrons.
- 9.
- Which of the following elements has the greatest attraction for electrons in a covalent bond?
- (a) Ge
- (b) As
- (c) Se
- (d) Br
- (e) Bi
- 10.
- Which statement is wrong?
- (a) The atomic weight of carbon is about 12.
- (b) The most stable ion of lithium is Li+.
- (c) A phosphorus atom is larger than an antimony atom.
- (d) The radius of a sodium atom is larger than that of a sodium cation.
- (e) Oxygen has a less negative electron affinity than fluorine.
- 11.
- All of the following properties of the alkaline earth metals increase going down the group except
- (a) atomic radius
- (b) first ionization energy
- (c) ionic radius
- (d) atomic mass
- (e) atomic volume
- 12.
- Which of the following is an ionic hydride?
- (a) PH3
- (b) H2S
- (c) HI
- (d) KH
- (e) CH4
- 13.
- Which of the following is the most basic oxide?
- (a) N2O3
- (b) N2O5
- (c) P4O6
- (d) P4O10
- (e) Bi2O5
Answers:
1. (d) 2. (a) 3. (c) 4. (c) 5. (c) 6. (e) 7. (d) 8. (b) 9. (d) 10. (c) 11. (b) 12. (d) 13. (e)

 Click here to return to the top.
Click here to return to the top.

 Choose your next chapter:
Choose your next chapter:

 To report any corrections, please e-mail Dr. Wendy Keeney-Kennicutt.
To report any corrections, please e-mail Dr. Wendy Keeney-Kennicutt.


 Li+(g) + e-
Li+(g) + e-
 Click here to return to the top.
Click here to return to the top.
 Choose your next chapter:
Choose your next chapter: 
 To report any corrections, please e-mail Dr. Wendy Keeney-Kennicutt.
To report any corrections, please e-mail Dr. Wendy Keeney-Kennicutt.