|


Department of Chemistry Texas A&M University PO Box 30012 College Station, TX 77842-3012 (t) 979.845.0215 (l) 979.845.4732 (f) 979.847.8660 |
Zirconium Clusters Synthetic exploration of halide clusters from the group IV and V transition metals, namely, Zr, Hf, Nb, and Ta, has lead to many novel materials with rich structural diversity and redox chemistry. The cluster motif most commonly observed in these compounds involves a centered metal octahedron with edge-sharing halide atoms and an electron deficient interstitial atom extending over three dimensions. Excision of the clusters from solids to obtain discrete molecules has lead to interesting research. Older work in Nb and Ta chemistry has been extended to Zr clusters by our group. One focus now is the use of these clusters as building blocks to form multi-dimensional materials through solution coordination chemistry. The reassembly of these discrete clusters will make materials that exhibit interesting optical, electrical and magnetic properties. The octahedral disposition of the outer coordination on these clusters is analogous to that of an octahedral metal atom and, therefore, lends itself as a good template for supramolecular assemblies. One challenge in the synthesis of supramolecular inorganic materials lies in the lack of control on the self-assembly process: lattice interpenetration and fast reaction rates are hard to prevent and lead to amorphous products. The differentiation of the coordination sites around the cluster core with chelating ligands will allow for lower dimensional assemblies in a more controlled manner. This project aims at the synthesis of kinetically inert zirconium halide cluster chelate complexes with bites of ~6 Å and their use as synthons in supramolecular assemblies. Specifically, we have been derivatizing 1,10-phenanthroline with various phosphine oxides to allow for adequate chelation of the zirconium cluster. The study of this interaction is underway and the results have been encouraging. 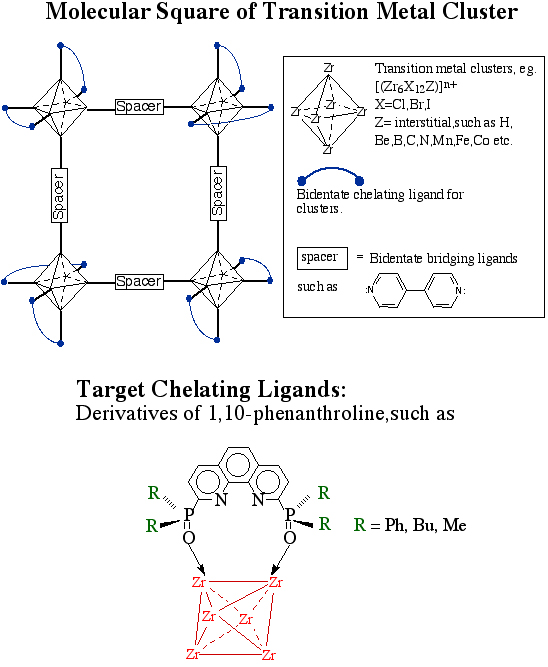 Qualitative DFT Study of the Interstitial NMR NMR studies of interstitial atoms nestled in zirconium halide clusters have yielded much to improve our understanding of the clusters in both solution and solid state. Compared with "ordinary" diamagnetic molecules, interstitial atom nuclei are highly deshielded. The deshielding increases as the bridging halide progresses from Cl to I. Work already completed provides the basis of a qualitative analysis of chemical shifts and gives insight into cluster bonding. Deshielding of interstitial atoms is attributable to the paramagnetic contribution (s
p), which arises from the second-order mixing of paramagnetic excited states into the ground state by the applied magnetic field. This paramagnetic contribution (s
Based on the Ramsay’s equation, we have correlated the chemical shifts for several nuclei (Z = 9Be, 11B, 13C) with {[(Zr6Z)X12]L6}n cluster bonding-antibonding energy gaps. A close examination of the MOs of the halide clusters with second row main group interstitials has focused our attention to the bonding (t1u) and antibonding (t1u*) orbitals resulting from the interaction between a Zr cage bonding orbital and the interstitial 2p orbital. (Scheme 1) Recently, we have performed DFT electronic structure calculations on a series of 11B and 13C centered zirconium clusters with different terminal ligands and bridging halides. Proportionality was found between the chemical shifts and the calculated energy gap D
|
|
(Updated: 8/30/2002) |
E-Mail Professor Hughbanks: trh@mail.chem.tamu.edu |


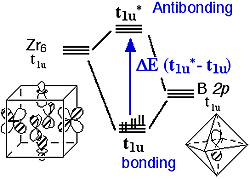 p) becomes significant only when both excited and ground states have a large interstitial atom orbital contribution and the right symmetry to allow for mixing. As a result of second-order mixing, the magnitude of paramagnetic contributions to the chemical shift is inversely dependent on the energy gaps between the ground and excited states.
p) becomes significant only when both excited and ground states have a large interstitial atom orbital contribution and the right symmetry to allow for mixing. As a result of second-order mixing, the magnitude of paramagnetic contributions to the chemical shift is inversely dependent on the energy gaps between the ground and excited states.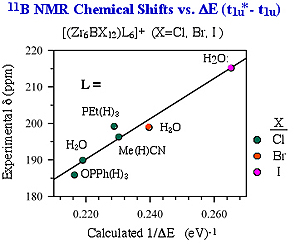 E-1(t1u-t1u*) for each series.(Figure 2) Therefore, we verified the most significant paramagnetic contribution of the interstitial chemical shifts. The trend of chemical shifts with the progression of bridging halides has been explained unambiguously by the weakening of bonding strength between the zirconium cage to the interstitial as the cluster core expands. Our calculation also shows that cluster core with stronger terminal ligand has greater D
E(t1u-t1u*), thus less deshielded.
E-1(t1u-t1u*) for each series.(Figure 2) Therefore, we verified the most significant paramagnetic contribution of the interstitial chemical shifts. The trend of chemical shifts with the progression of bridging halides has been explained unambiguously by the weakening of bonding strength between the zirconium cage to the interstitial as the cluster core expands. Our calculation also shows that cluster core with stronger terminal ligand has greater D
E(t1u-t1u*), thus less deshielded.