|


Department of Chemistry Texas A&M University PO Box 30012 College Station, TX 77842-3012 (t) 979.845.0215 (l) 979.845.4732 (f) 979.847.8660 |
Extensive synthetic investigation of metal-rich materials has lead to a great variety of new chemistry: compounds with novel compositions, structures, and properties continue to be discovered. Following the discovery of a series of Zr- and Hf-rich chalcogenides in our group, novel rare earth metal-rich telluride compounds incorporating late transition metals have been investigated.
Gd6MTe2 (M = Co, Ni) and Er6RuTe2 crystallize in the Zr6CoAl2-type structure. Three-fold symmetric tricapped trigonal prismatic clusters, each of which is centered with a late transition metal, are the basic building blocks. Most conspicuous in the structure are the metal-metal-bonded chains constructed by condensation of such cluster units. The chains in turn interconnect to yield an extended structure in three dimensions. The Zr6CoAl2-type structure exhibits great flexibility with respect to electron counts and element sizes. Compounds isotypic to Er7M2Te2 (M = Co, Ni) have not been found yet for any other metals in Groups III, IV, and V.
The major difference between Er5M2Te2 (M = Co, Ni) and those mentioned above is the presence of M-M bonding in the former. The structural motifs are monocapped trigonal prisms of Er with late transition metals located near the center. These units are fused by face-sharing such that the late transition metals (M) are in relatively close proximity. Similar structural features have been observed in Sc5Ni2Te2 and Gd3MnI3. We are studying the magnetic properties of quasi-molecular rare earth halide cluster compounds, where the cluster units are isolated from each other, in hopes of introducing a new and promising class of rare earth-containing compounds into single-molecule magnet research.
From a theoretical point-of-view, there exists a need for chemically useful yet physically realistic bonding schemes that can help us construct interpretive and predictive magnetostructural models for molecule-based lanthanide systems. Important criteria for an accurate descriptive model include providing good semiquantitative estimates for the exchange coupling constants (J's) that account for observed ferro- or antiferromagnetic properties, and correlating the magnetostructural properties within a series of compounds. Our goal is to provide a basis of correlating structure, bonding, and magnetic properties with more specific applicability than the more phenomenological RKKY description. Exploiting intraatomic exchange between 4f and 5d electrons in lanthanide systems is key to understanding and, ultimately, to designing rare-earth molecular systems with large magnetic moments at high temperature. To design such compounds synthetically, cation substitution is a chemical alternative to simplify the magnetic properties of the R(R6X12Z) compounds. Gd(Gd6I12Z) cluster compounds exhibit magnetic properties that arise from two types of Gd ions (isolated Gd3+ and metal-metal bonded Gd). The isolated Gd3+ ions (4f7) are fully oxidized and are likely to be insignificantly coupled to neighboring magnetic moments (i.e. they have localized S = 7/2 spins). In contrast, the Gd atoms within the Gd6 clusters are bonded to each other and to the central Z atom. The highest occupied orbitals of the clusters are mainly of Gd 5d character. It is our hypothesis that when unpaired electrons are present in the cluster, intraatomic exchange interactions will align each 4f moment with the unpaired 5d spin, resulting in the ferromagnetic alignment of all six 4f 7 moments. If the Gd3+ can be successfully replaced with a non-magnetic cation, then the magnetic properties of the material should reflect those of the Gd6 clusters alone. A long-term goal of this research is to synthesize solids where clusters are more structurally isolated and/or to excise them into solution is of great challenge. Of currently known compounds, high-spin cluster units are highly crosslinked with each other in the solid state. Nevertheless, cluster excision may be an ultimate chemical solution to clarifying the intracluster magnetic coupling mechanism and to exploiting this new class of high-spin molecules. |
|
(Updated: 8/30/2002) |
E-Mail Professor Hughbanks: trh@mail.chem.tamu.edu |


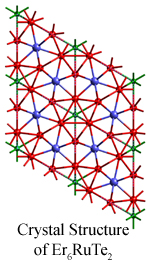 Ternary chalcogenides with surprisingly simple compositions have challenged chemist's conventional understanding of metallic bonding. Strong bonding between the rare earth and the late transition metals is a key factor favoring the formation of such compounds. Stabilization by polar intermetallic bonding has been practiced as an efficient synthetic strategy in the exploration of these new metal cluster compounds.
Ternary chalcogenides with surprisingly simple compositions have challenged chemist's conventional understanding of metallic bonding. Strong bonding between the rare earth and the late transition metals is a key factor favoring the formation of such compounds. Stabilization by polar intermetallic bonding has been practiced as an efficient synthetic strategy in the exploration of these new metal cluster compounds. 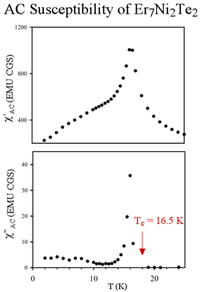 Once again, the tricapped trigonal prismatic building block is easily recognized in the structure. These cluster units condense by sharing faces, edges, and/or vertices to form chains, layers and finally 3D networks. The Er7M2Te2 structure is more metal-rich than others, as characterized by its more condensed metal framework. Band structure calculations using the extended Hückel method indicate the material is metallic, as suggested by its metal-metal-bonded framework. Magnetic measurements on the polycrystalline sample of Er7Ni2Te2 reveal paramagnetic behavior consistent with Er3+ cores, but an ordering transition occurs at 16.5 K. This is the first rare earth metal-rich ternary chalcogenide of which a magnetic transition has been observed.
Once again, the tricapped trigonal prismatic building block is easily recognized in the structure. These cluster units condense by sharing faces, edges, and/or vertices to form chains, layers and finally 3D networks. The Er7M2Te2 structure is more metal-rich than others, as characterized by its more condensed metal framework. Band structure calculations using the extended Hückel method indicate the material is metallic, as suggested by its metal-metal-bonded framework. Magnetic measurements on the polycrystalline sample of Er7Ni2Te2 reveal paramagnetic behavior consistent with Er3+ cores, but an ordering transition occurs at 16.5 K. This is the first rare earth metal-rich ternary chalcogenide of which a magnetic transition has been observed.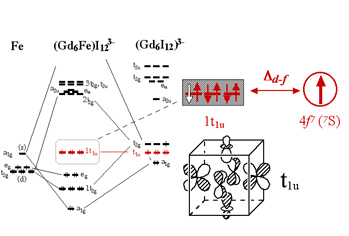 Preliminary results indicate that discrete clusters possessing unpaired electrons in Ln-Ln bonding molecular orbitals may exhibit strong intramolecular ferromagnetic coupling. Considering the complexity of the solid-state materials and the subject of magnetism, it is challenging to establish unambiguous evidence for magnetic ordering within clusters.
Preliminary results indicate that discrete clusters possessing unpaired electrons in Ln-Ln bonding molecular orbitals may exhibit strong intramolecular ferromagnetic coupling. Considering the complexity of the solid-state materials and the subject of magnetism, it is challenging to establish unambiguous evidence for magnetic ordering within clusters. 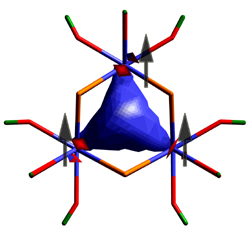 The models help us study interatomic exchange, mediated by spin polarization of both filled and partially-filled bonding orbitals, using perturbation methods. This provides semiquantitative estimates of the energetic ordering of competing spin states. Using fragments based on metal-rich lanthanide solids, we developed polynuclear Gd cluster models that exhibit magnetic moment coupling resembling that of its solid-state "parent." Detailed DFT calculations were used to construct and check features of a generally applicable qualitative approach to understanding magnetic coupling in rare-earth-rich compounds.
The models help us study interatomic exchange, mediated by spin polarization of both filled and partially-filled bonding orbitals, using perturbation methods. This provides semiquantitative estimates of the energetic ordering of competing spin states. Using fragments based on metal-rich lanthanide solids, we developed polynuclear Gd cluster models that exhibit magnetic moment coupling resembling that of its solid-state "parent." Detailed DFT calculations were used to construct and check features of a generally applicable qualitative approach to understanding magnetic coupling in rare-earth-rich compounds.