


Students in Texas A&M's 2023 “Smash Chemistry” STEM camp use a luminescent polymer to study how forces are distributed in a mechanochemical reactor. | Image: Courtesy of Dr. James D. Batteas
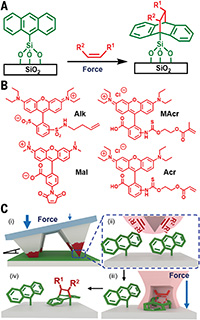
Fig. 1. Force-activated Diels-Alder reactions on anthracene monolayers.
(A) Diels-Alder cycloaddition reaction between anthracenes immobilized onto SiO2 surfaces and dienophiles under applied force. (B) Structures of dienophiles Alk, Mal, MAcr, and Acr. (C) (i) Elastomeric tip arrays transfer an ink mixture (red coating), consisting of a dienophile and PEG, onto an anthracene-modified surface. The tip arrays are tilted so that different forces are applied by the tips at different locations across the surface. Thick arrows indicate areas of the tip array that exert high force, and thin arrows indicate areas that exert low force. (ii) Upon contact with the surface, the tips form nanoreactors, (iii) where forces are applied that accelerate the Diels-Alder cycloaddition reactions. (iv) After washing the surface, only covalently bound molecules remain on the surface
Kueyoung Kim's poster presentation at the Penn State's Eberly College of Science Undergraduate Poster Session represents his work this summer studying mechanochemistry at the University of Pennsylvania with Professor R.W. Carpick and postdoc Dr. Pranjal Nautiyal.
Kueyoung Kim (3rd person from the right)
Mechanochemistry dates back to antiquity, and is one of the oldest forms of carrying out chemical transformations. The application of force by the mechanical grinding and mixing of materials helps drive chemical reactions.
Recently mechanochemistry has been seeing a major resurgence due to the increased interest in developing greener and more sustainable means for doing chemistry.
This digital exhibit is the first of a series being developed to explore the history and future of mechanochemistry. We give you a snapshot into the historical origins of mechanochemistry as well as a sneak peek into where it is going. We hope you enjoy it!