The vision of the NSF Center for the Mechanical Control of Chemistry (CMCC) is to establish a fundamental understanding of mechanochemistry by combining new instrumental, chemical, and theoretical approaches, thereby enabling the robust mechanical control of chemical reactions for the design, prediction, and scale-up of sustainable materials synthesis.
The mission of the center is to serve as a locus for the integration of research, education, innovation, and outreach for the mechanochemistry community and beyond. The CMCC will develop and provide new experimental and theoretical toolsets for the fundamental study of mechanochemical reactions across a range of chemical systems and, in collaboration with industry, will translate this fundamental understanding and tools into practice to realize the goals of sustainable chemical synthesis, broadly defined. The center will actively promote workforce development in sustainable chemical manufacturing through the training of the next generation of mechanochemists.
The NSF Center for the Mechanical Control of Chemistry (CMCC) is an integrative research and training environment that brings together chemists, physicists, and engineers around the common goal of establishing a fundamental understanding of mechanochemistry—the use of mechanical force to drive chemical reactions. Mechanochemistry offers a powerful and versatile means for chemical synthesis, opening the door to new reactions and new materials that cannot be made using traditional synthetic methods. While the effects of light, heat, and electric charge for driving chemical reactions are well understood, the use of mechanical force as a controlling synthetic element is far less developed. To unveil the full potential of mechanochemistry, the CMCC aims to bridge key knowledge gaps between the fields of chemistry and mechanics which have limited the broad application of mechanochemistry as a core synthetic methodology. As mechanochemical reactions can often be carried out with little or no solvent, and at lower temperatures than traditional synthetic approaches, the translation of knowledge in the CMCC from molecular-scale understanding, to large scale syntheses, has the potential for major technological and economic benefits globally, affording more sustainable approaches to the synthesis of chemicals and materials.
Why should the world care about mechanochemical reactions, when we have perfectly good thermally, optically, and electrically-driven chemical reactions?
One big reason is the potential for eliminating the need for costly, toxic, and wasteful solvents. Industrial chemical synthesis is a multi-trillion dollar industry that globally represents 7% of all income, producing nearly a billion tons of products each year. Within the ~ $1T sub-sector of specialty chemicals, reactions are largely carried out in liquid solvents, requiring upwards of 30 tons of solvent for a 1 ton yield of useful product, resulting in over 5B gallons/yr of costly and toxic solvent waste. Timely efforts in mechanochemical synthesis have demonstrated that many high-value chemicals can be synthesized with little or no solvent, sometimes with even higher yield than conventional synthetic methods! Thus, improving our understanding of mechanochemistry will provide monumental improvements in the economics and environmental impact of the chemical industry.
Beyond the impacts to chemical synthesis, mechanochemistry lies at the very heart of tribochemistry, which underpins the key energy loss and failure mechanisms at interfaces in sliding contact. In passenger vehicles, frictional losses in components consume 28% of fuel energy, and 66% of the energy in electricity generation is similarly lost to conversion inefficiencies. Design of mechanochemically-active lubricant additives is key to reducing friction and wear, which contributes to losses of ~$200 billion/year in the US alone. Thus, over its 13-year lifetime, CMCC’s findings will not only alter the vast chemical synthesis landscape, but could result in manifold improvements in energy efficiency.
In addition to fundamental research, the CMCC will also be establishing a Mechanochemistry Innovation Hub, to serve as a nexus, for pairing industry (and other interested stakeholders) with CMCC researchers to explore the feasibility of mechanochemical methods for adaption to critical processes.
(Leads: Dr. Jonathan Felts and Dr. Andrew Rappe)
(Leads: Dr. Adam Braunschweig and Dr. James Mack)
(Leads: Dr. Danna Freedman and Dr. James Rondinelli)
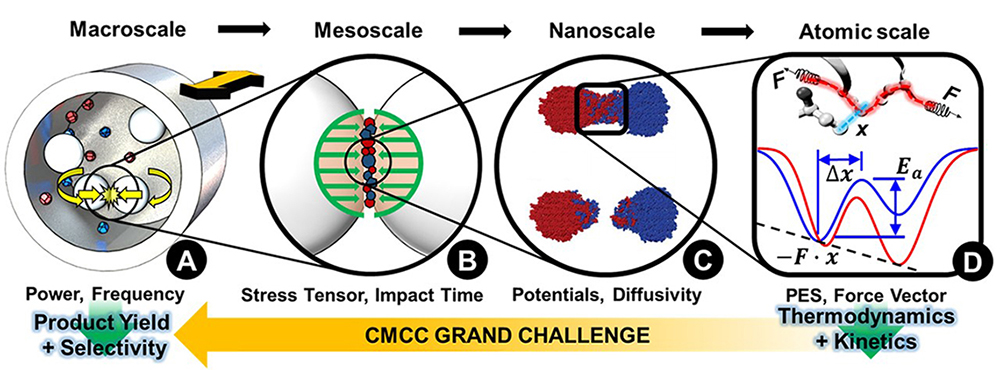
In both RTs, starting at the nanoscale, we will examine reactions at well-defined interfaces, where force vectors and molecular orientations can be controlled and/or characterized. These studies will drive hypotheses elucidated by simulations, theory, and data-driven approaches to explain the key factors controlling mechanochemistry, including kinetics, functional group mechano-susceptibility, and stereoselectivity. These hypotheses will in turn be tested against experimental results including reactions run in novel mechanochemical reactors. Cross-fertilization between the ITP and both RTs will be fostered to lead to a robust understanding of the factors that dictate critical reaction parameters. Through this structure, we will address the CMCC’s grand challenge to scale up atomistic mechanochemistry theory to enable predictive macroscale synthesis.