D. J. Darensbourg - Research Laboratories

Current Research
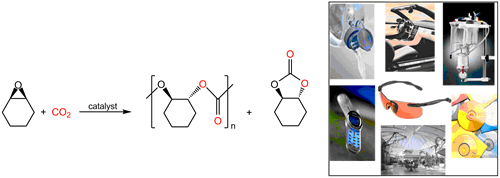
Prominent among the processes that represent technologically viable uses of carbon dioxide, both as a solvent and a monomer, is the production of biodegradable copolymers. Although carbon dioxide is thermodynamically and kinetically very stable, it does undergo reactions with highly reactive molecules, most notably for our purposes herein are transformations with ring-strained heterocycles such as propylene oxide and cyclohexene oxide (Scheme 1). As noted in Scheme 1, these processes are generally accompanied by the production of differing amounts of five-membered cyclic carbonates, with aliphatic epoxides affording larger quantities than alicyclic epoxides. Of importance, both the copolymer and its corresponding cyclic carbonate afforded from the alternating coupling of epoxides and carbon dioxide have industrial applications. That is, polycarbonates are known for their outstanding properties, including strength, lightness, durability, high transparency, heat resistance and good electrical insulation. Cyclic carbonates find uses as high boiling solvents and reactive intermediates.
SCHEME 1
An alternative source of polycarbonate derived from carbon dioxide involves the alternating copolymerization of oxetane and carbon dioxide (eq 1). The ring-strain energy of oxetane is less than that of propylene oxide, 106.7 kJ/mol vs 114.2 kJ/mol, hence its copolymerization with carbon dioxide is less favored thermodynamically. Nevertheless, the copolymerization of oxetane and carbon dioxide readily occurs under similar catalytic conditions, producing poly(trimethylene carbonate), an important component of biodegradable thermoplastics elastomers which have numerous clinical applications. A significant difference between this coupling reaction and the epoxide/CO2 process is that in this instance the six-membered cyclic carbonate is thermodynamically unstable relative to the copolymer.
.png)
EQ. 1
Current research efforts in this program are aimed at improving the activity and selectivity of these metal catalysts for the production of polycarbonates from both epoxides and four-membered cyclic ethers(oxetanes). Importantly, the polymers produced from oxetanes and CO2 when coupled with polyesters provide biodegradable thermoplastic elastomers which have various applications in biomedical areas such as surgical sutures, drug delivery devices, and body or dental implants. Relative to this latter subject, research is being directed at developing catalysts derived from biocompatible metals, specifically calcium and zinc, for producing these materials from trimethylene carbonate and lactides which are obtained from corn.