Watanabe Research Lab


The azinomycins are anti-tumor antibiotics isolated from the culture broths of the terrestrial microorganism, Streptomyces sahachiroi. Their biological activity appears to reside in their ability to covalently alkylate and subsequently cross-link double stranded DNA.
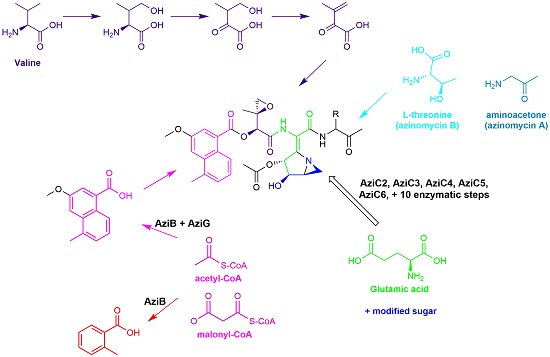
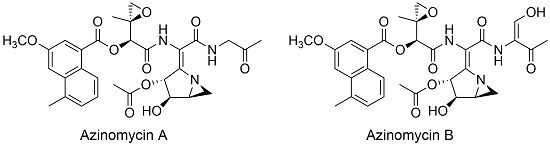
Our research group is actively characterizing the biosynthetic genes of this pathway, which involves a variety of techniques and strategies including: cloning and overexpression of genes, disruption/knockout of genes, enzymology, as well as chemical synthesis/isotopic labeling studies. Functional characterization of the genes of the pathway will not only shed light on the mechanism of azabicycle formation but will also pave the way for genetic engineering of the pathway and the development of new therapeutic methodologies.
We have also been investigating the biosynthesis and cellular effects of cycloterpenals and their derivatives. Cycloretinal (all-trans retinal dimer), a representative member of this family of natural products is attributed to causing age-related macular degeneration (AMD). AMD is the leading cause of blindness in adults over the age of 50 that can lead to the loss of central vision.
One of the most common early characteristic features of AMD (the dry form) is the accumulation of yellow deposits in the eye called drusen. A more severe form of the disease, the wet form, is characterized by neovascularization (abnormal blood vessel formation). Cycloretinal is a non-degradable, toxic byproduct of the visual cycle. Photochemical activation of the metabolite leads to oxidative damage of the retinal pigment epithelium and adjacent photoreceptor cells.
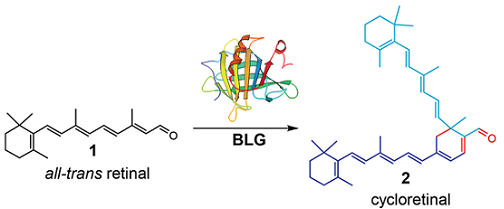
Biochemical evidence from our lab has recently revealed that beta-lactoglobulin (BLG), the principal whey protein in milk, biosynthesizes the lipofuscin cycloretinal (all-trans retinal dimer). The protein survives the pasteurization process, is acid stable and a large fraction of the protein is transported intact through the blood stream (as supported by rabbit studies). Proteomic studies performed by Crabb and co-workers have shown that the protein is present in drusen.
US Mail
Department of Chemistry
Texas A&M University
PO Box 30012
College Station, TX
77842-3012
Express Shipping
Department of Chemistry
Texas A&M University
Ross St @ Spence St
College Station, TX 77843-3255
Campus Mail
Department of Chemistry
TAMU 3255
College Station, TX 77843-3255