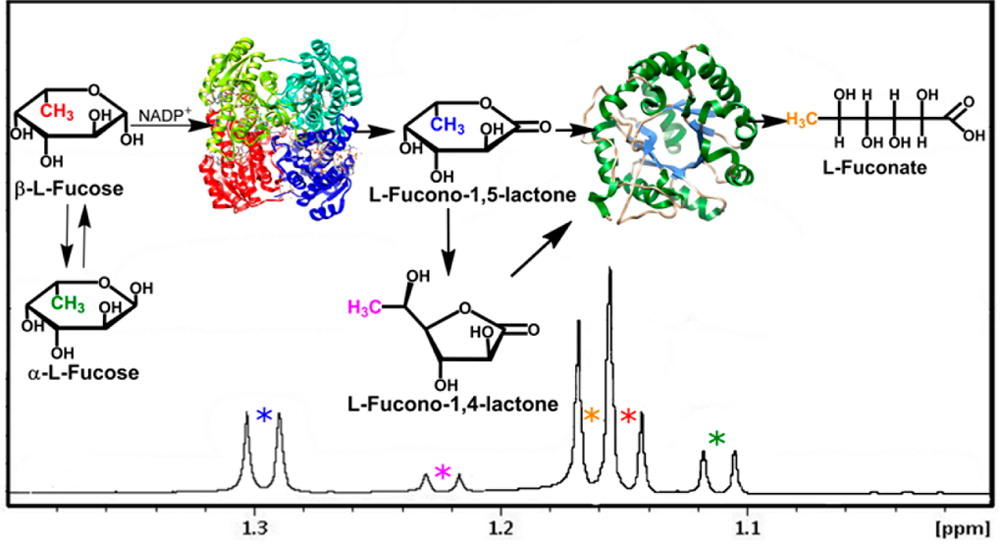
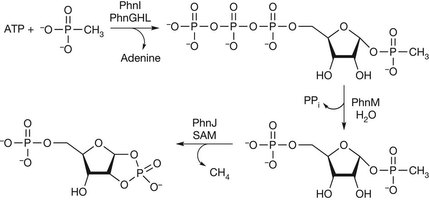
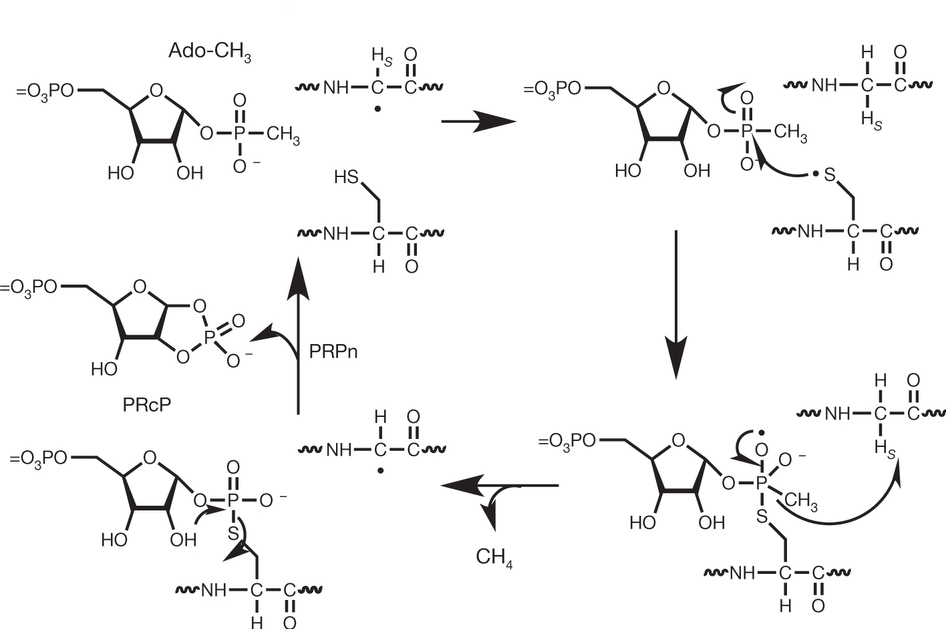
Our research efforts are directed towarad a more complete understanding of the fundamental principles involved in enzyme-catalyzed chemistry and protein structure. The acquisition of this information is providing the framework for the redesign of these complex molecules in a concerted effort to exploit the properties of enzyme active sites for a variety of chemical and medicinal uses. The techniques that we are using to solve these problems include steady-state and stopped-flow kinetics, nuclear magnetic resonance spectroscopy (NMR), electroparamagnetic resonance (EPR), x-ray crystallography, chemical synthesis of inhibitors and novel substrates, and site-directed mutagenesis for the construction of new proteins with unique catalytic properties.