Development of (NO)Fe(N2S2) as a Metallodithiolate Spin Probe Ligand: A Case Study Approach
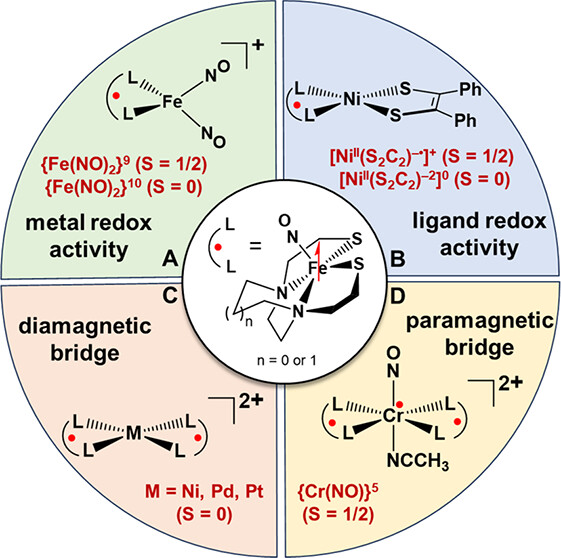
(M282) Manuel Quiroz and Marcetta Darensbourg. Acc. Chem. Res., 2024, 57, 831-844.
|
Metal‐Templated, Tight Loop Conformation of a Cys‐X‐Cys Biomimetic Assembles a Dimanganese Complex
(M269) T. Le, H. Nguyen, L. M. Perez, D. J. Darensbourg, M. Y. Darensbourg. Angew. Chem. Int. Ed., 2020, 59, 2-7.
|
Controlling O2 Reactivity in Synthetic Analogues of [NiFeS]- and [NiFeSe]-Hydrogenase Active Sites
(M267) X. Yang, L. C. Elrod, T. Le, V. S. Vega, H. Naumann, Y. Rezenom, J. H. Reibenspies, M. B. Hall, M. Y. Darensbourg. J. Am. Chem. Soc. 2019, 141, 15338-15347.
|
Toward the Optimization of Dinitrosyl Iron Complexes as
Therapeutics for Smooth Muscle Cells

(M265) D. C. Pectol, S. Kahn, R. B. Chupik, M. Elsabahy, K. L. Wooley, M. Y. Darensbourg,
S.-M. Lim. Mol. Pharmaceutics. 2019, 16(7), 3178 - 3187.
|
Oxygen uptake in complexes related to [NiFeS]- and [NiFeSe]-hydrogenase active sites
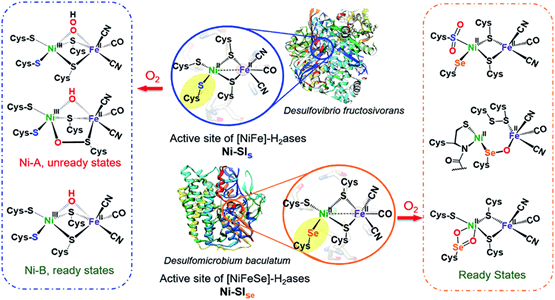
(M264) X. Yang, L. C. Elrod, J. H. Reibenspies, M. B. Hall, M. Y. Darensbourg. Chem. Sci. 2019, 10, 1368 - 1373.
|
Bridging cyanides from cyanoiron metalloligands to redox-active dinitrosyl iron units
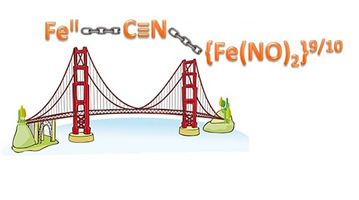
(M260) P. Ghosh, M. Quiroz, R. Pulukkody, N. Bhuvanesh,
M. Y. Darensbourg. Dalton Trans. 2018, 47, 11812 - 11819.
|
Interplay of hemilability and redox activity in models of
hydrogenase active sites
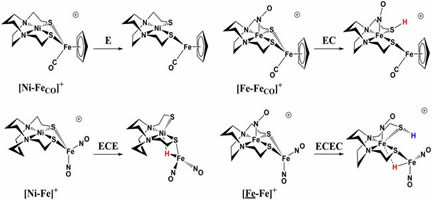
(M258) S. Ding, P. Ghosh, M. Y. Darensbourg, M. B. Hall. PNAS,
2017,114 (46), E9775-E9782.
|
Cyanide Docking and Linkage Isomerism in Models for the Artificial [FeFe]-Hydrogenase Maturation Process
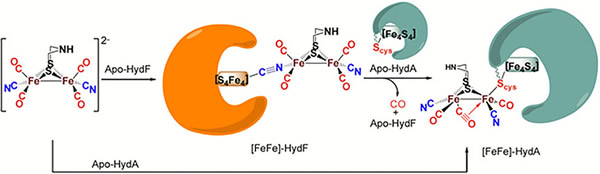
(M261) D. Sil, Z. Martinez, S. Ding, N. Bhuvanesh, D. J. Darensbourg, M. B. Hall, M. Y. Darensbourg. J. Am. Chem. Soc. 2018, 140 (31), 9904–9911.
|
Complexes of MN2S2•Fe(η5-C5R5)(CO) as Platform for Exploring Cooperative Heterobimetallic Effects in HER Electrocatalysis
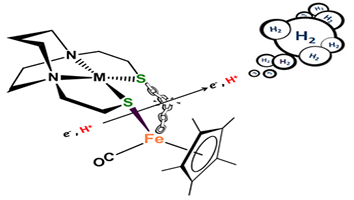
(M255) P. Ghosh; M. Quiroz; N. Wang; N. Bhuvanesh; M. Y. Darensbourg,
Dalton Trans. 2017, 46, 5617 - 5624. |
Discrete Air-Stable Nickel-Palladium(II) Complexes as
Catalysts for Suzuki-Miyaura Reactions
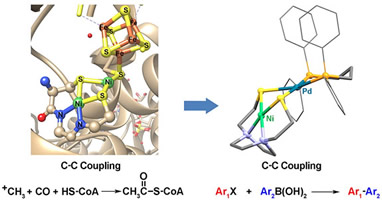
(M256) T. Zhao; P. Ghosh; Z. Martinez; Xu. Liu; X. Meng; M. Y. Darensbourg,
Organometallics, 2017, 36, 1822–1827. |
Hemi-labile Bridging Thiolates as Proton Shuttle in
Bio-inspired H2 Production Electrocatalysts
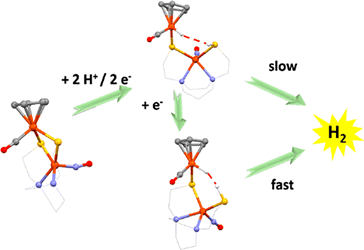
(M252) S. Ding, P. Ghosh, A. M. Lunsford, N. Wang, N. Bhuvanesh, M. B. Hall,
M. Y. Darensbourg, J. Am. Chem. Soc. 2016, 138 (39), 12920–12927.
|
Toward Biocompatible Dinitrosyl Iron Complexes:
Sugar-Appended Thiolates
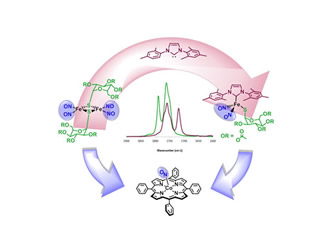
(M253) R. Pulukkody, R. B. Chupik; Steven K. Montalvo; Sarosh Khan; Nattamai Bhuvanesh; Soon-Mi Lim; Marcetta Y. Darensbourg, Chem. Commun. 2017, 53, 1180-1183.
|

